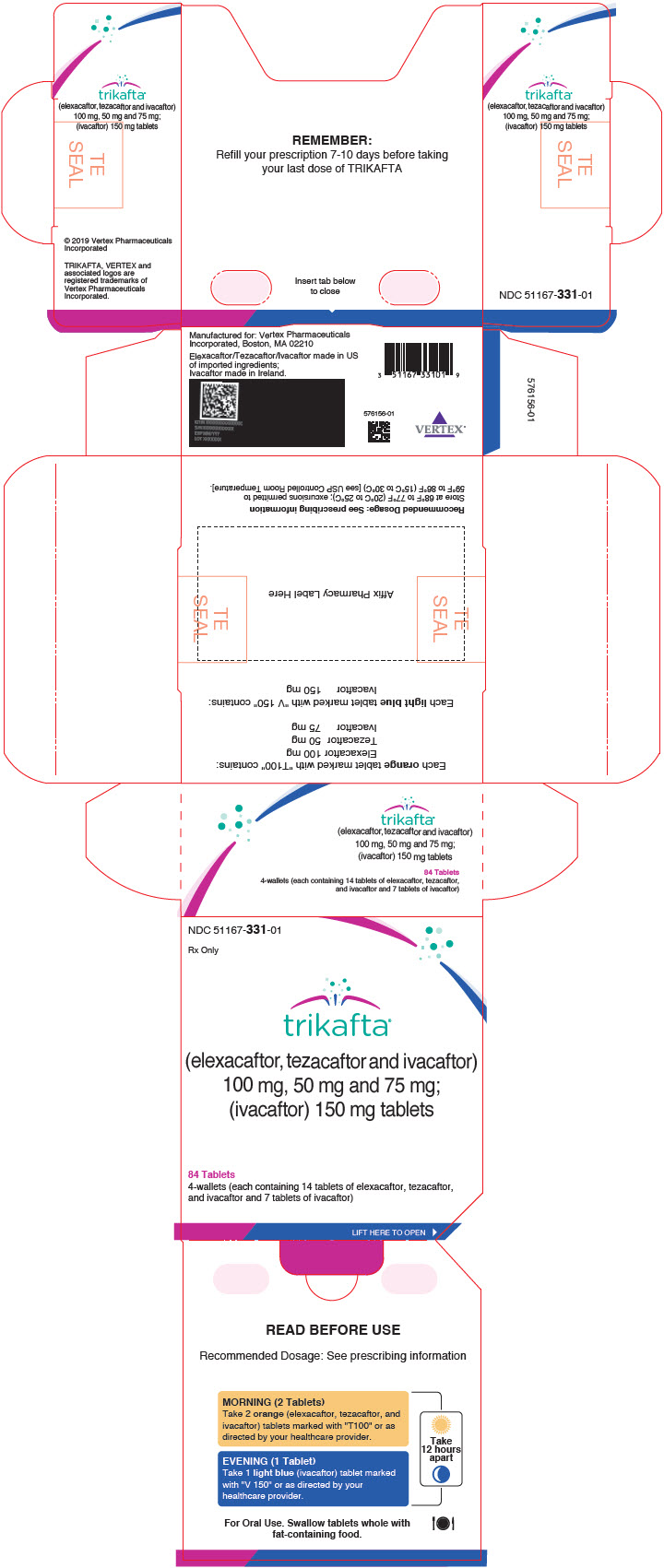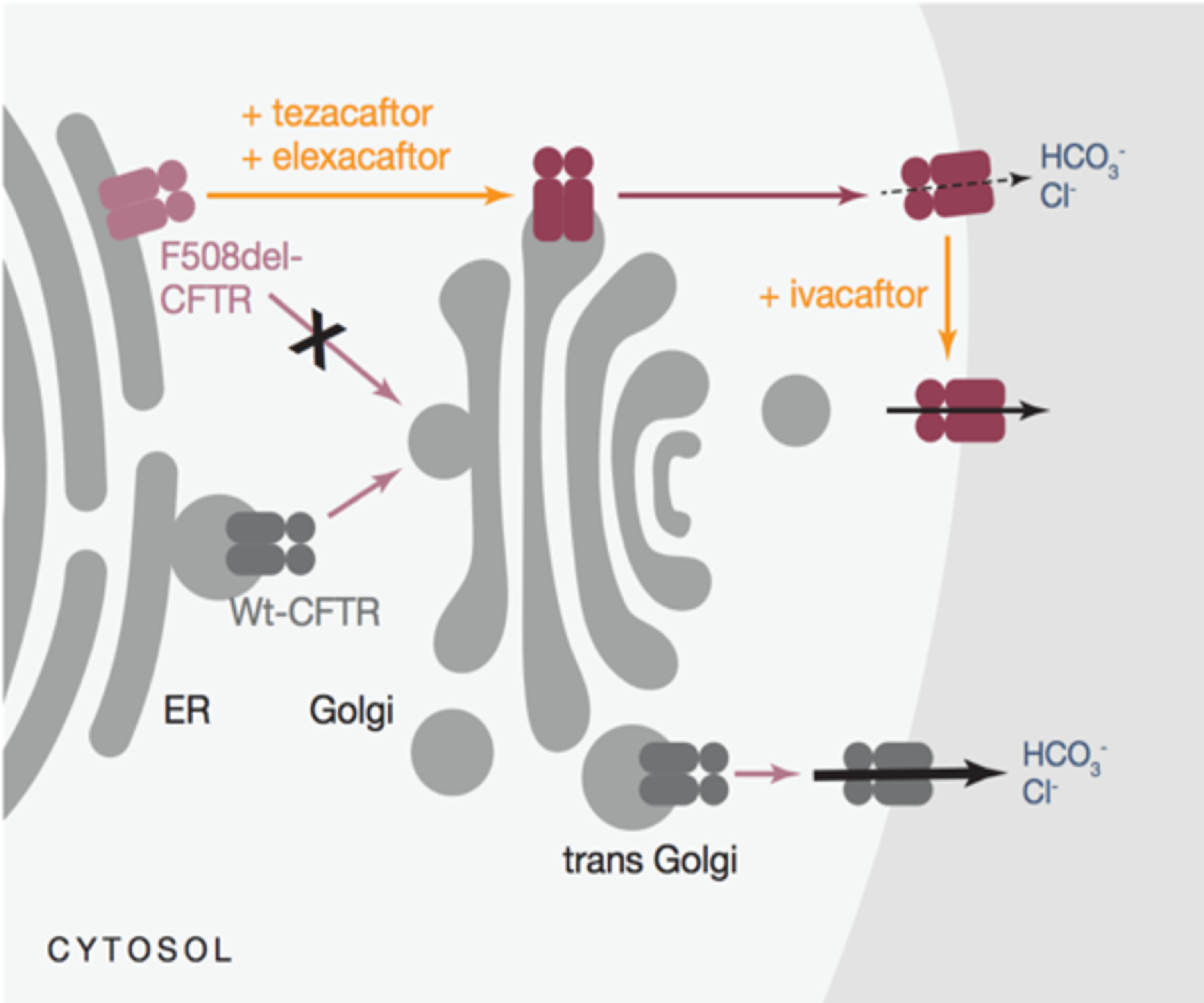
Cureus | A Review of Trikafta: Triple Cystic Fibrosis Transmembrane Conductance Regulator (CFTR) Modulator Therapy

Long-term safety and efficacy of tezacaftor–ivacaftor in individuals with cystic fibrosis aged 12 years or older who are homozygous or heterozygous for Phe508del CFTR (EXTEND): an open-label extension study - The Lancet
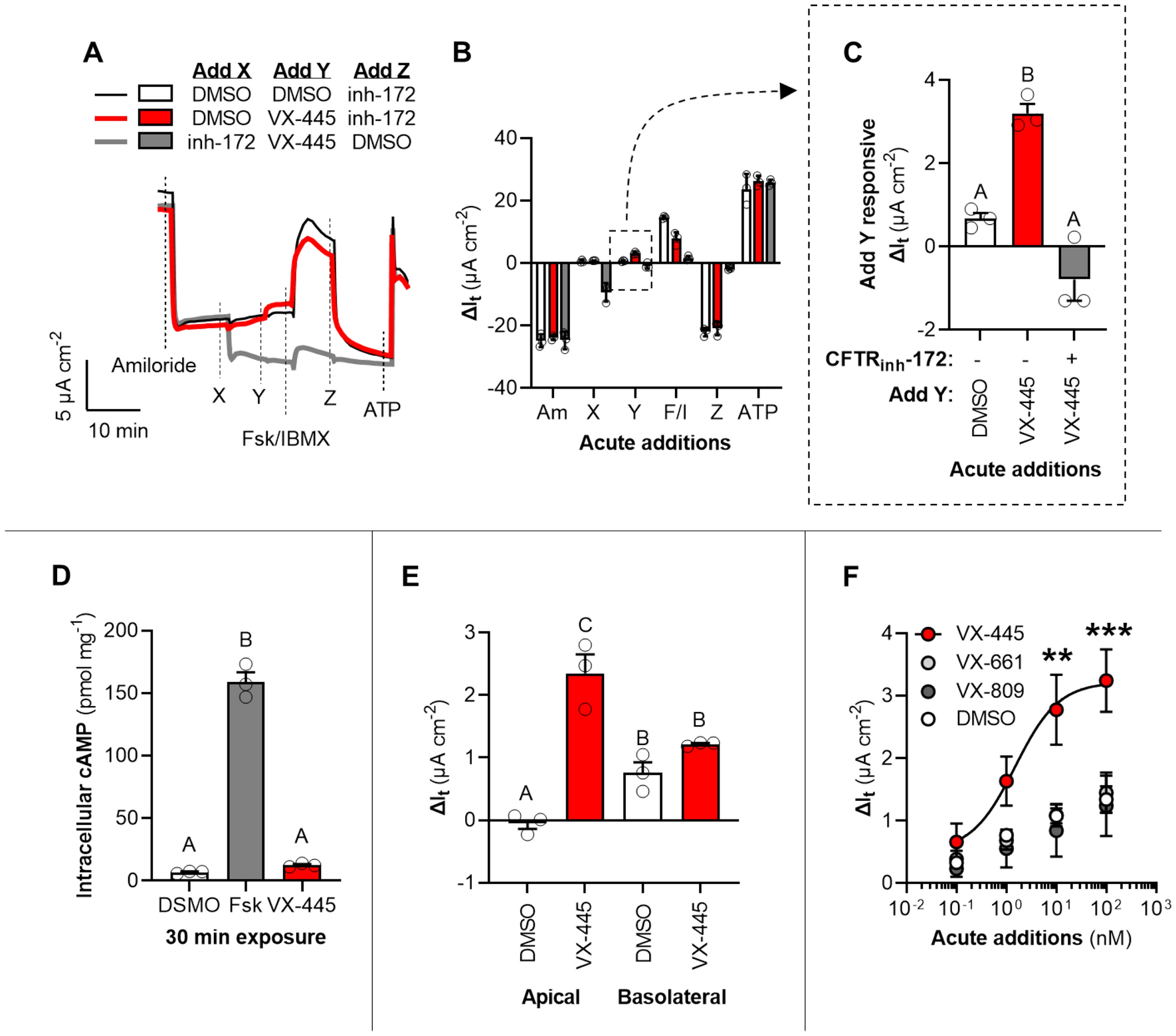
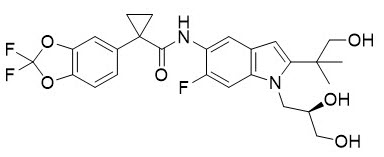
Elexacaftor is a CFTR potentiator and acts synergistically with ivacaftor during acute and chronic treatment | Scientific Reports

Share petition · Ministère de la sante: Accord France Vertex MAINTENANT contre la mucoviscidose · Change.org

NHS spent £100million on deal with a US pharmaceutical firm for the wrong cystic fibrosis drug | Daily Mail Online

Trikafta/Kaftrio continues to be a game-changer for cystic fibrosis patients - Pharmaceutical Technology

KaftrioNOW: The Buzz Tag For Cystic Fibrosis That Is Trending On Social Media | by Emma Boniface | Coughy and Creon | Medium