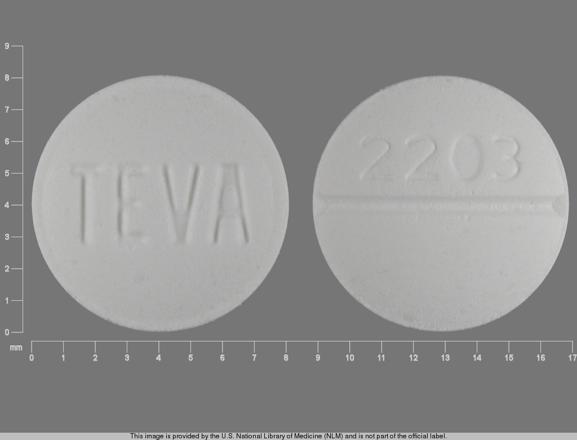
Efficacy and safety of betahistine treatment in patients with Meniere's disease: primary results of a long term, multicentre, double blind, randomised, placebo controlled, dose defining trial (BEMED trial) | The BMJ

PDF) Long-term prophylactic treatment of attacks of vertigo in Meniére's disease - Comparison of a high with a low dosage of betahistine in an open trial